14) Immunology
No studies are known to us, other than serologic surveys. Thus, Anderson & Rowe (1998) found antibodies to Rift Valley Fever (RVF) virus in black rhinos of Zimbabwe . Fischer-Tenhagen et al. (2000) found antibodies to a variety of other viral agents, but not against RVF virus in 281 animals from South Africa , Kenya and Namibia.
15) Pathological features
A major problem for black rhinoceroses is the propensity to develop hemolytic anemia (Chaplin, et al., 1986; Miller & Boever, 1982; Paglia et al., 1986; Paglia, 1993). The major enzymatic causes of this phenomenon were well described by Paglia (1993), and hematologic parameters studied by Chaplin et al. (1986). Veterinary problems, diseases and numerous procedures, were summarized by Silberman & Fulton (1979), and a complete veterinary bibliography was compiled by Miller (1983). It includes references to reproductive laboratory data as well.
Dermal problems and infections appear to be the commonest ailments, including pox (Schaller & Pilaski, 1979; Griner, 1983). Skin ulcers, studied by Kock & Kock (1990) in free-living animals from Zimbabwe were mostly or only due to filarial nematodes ( Stephanofilaria dinniki ). Other parasites have been described; thus, Keirans (1993) found Ixodid ticks in white and black rhinos; babesiasis was reported in three animals from Tanzania (Nijhof et al., 2003); trypanosomiasis was reported by Mihok et al. (1992) and Knapp et al. (1997) collected various arthropods and helminths from black rhinos. Neiffer et al. (2001) described leptospirosis in two black rhinos from Pittsburgh Zoo, presumably acquired from raccoons. Salmonellosis killed two rhinos in Denver (Kenny et al., 1997) and a survey of US zoos (Kenny, 1999) indicated an approximately 10% infection rate.
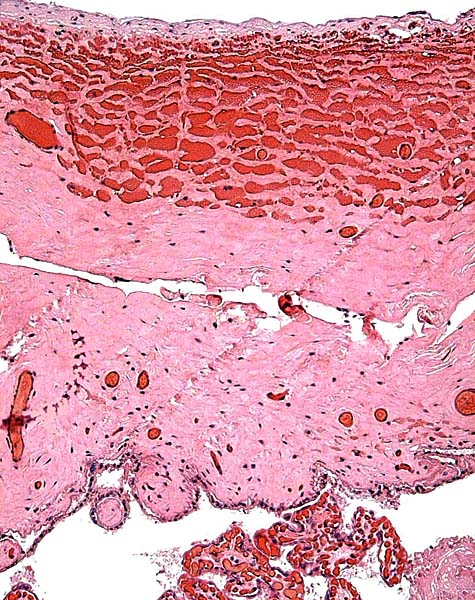
Three captive black rhinos showed severe encephalomalacia at death whose etiology remains unknown (Miller et al., 1990). Jones (1979) saw ulcerative stomatitis in black rhinos, a lesion later also described by Ott et al. (1982). Pessier et al. (2004) later associated this to eosinophilic granulomas occurring in rhinos but distinguished it from the necrolytic dermatitis described by Munson et al. (1998). Such skin lesions led Grant et al. (2002) to study fatty acids of the foods eaten; diets in the wild were significantly different in their composition. Following prolonged antibiotic therapy of hoof abscesses, a black rhino died from pulmonary aspergillosis (Woods et al., 1999). Acute lymphoblastic leukemia in a 21 months-old animal was the cause of death, perhaps also related to cardiotoxicity of the therapeutic agents (Radcliffe et al., 2000; Paglia & Radcliffe, 2000). That case study suggested excess iron in black rhinos, a notion that was more recently confirmed by additional studies (Paglia et al., 2001). These studies suggested greater prevalence of iron storage in females and suggested a link to the encephalopathy. Stegmann et al. (2001) described the fascinating story of anesthesia and operation of a necrotic rectal prolapse in a free-ranging black rhino. The animal died and was then found to have old pelvic fractures that may have been contributory (Olivier et al., 2001).
16) Physiologic data
Whatever relevant physiologic data are available have been provided in review form by Silberman & Fulton (1979), and as bibliography by Miller (1983). Schaurte (1966) described in detail the various dental formulas and depicts teeth of all rhinocerotidae. Schaffer et al. (1998) studied the reasons for the difficulty of obtaining semen by using sonography. They demonstrated the urethral filling in conditioned animals and the benefits of sonographic usage. O'Brien & Roth (2000) provided a protocol for the conservation and recovery of semen from Sumatran and black rhinos. Meiswinkel (1987) described mosquitoes grown from elephant and rhino dung. Schaffer et al. (2001) found the anatomy and histology of reproductive tracts similar to other species. Maluf (91991; 1994) described the anatomy of the kidney with its 60 lobes. Dierenfeld et al. (1988) found that plasma vitamin E levels of 31 wild animals were significantly higher than in eleven animals of the captive population. In a subsequent study, these results were not verified, presumably because of vitamin supplementation in captive animals (Clauss et al., 2002). Hematologic parameters, especially following the frequent translocations occurring in Africa, were delineated by Kock et al. (1999). The episodic occurrences of hemolysis have led to study particulars of enzymes etc. in rhino red cells. Thus, Weber et al. (2004) found significantly higher tyrosine levels in wild animals compared to captive specimens. For the same reasons, the hemoglobins were studied in several animals and related species (Fairbanks & Miller, 1990). They found considerable polymorphism of black rhino hemoglobins and proposed a genetic mechanism for these proteins.
17) Other resources
Cell strains of four species of rhinoceros are available from the “Frozen zoo” at the Zoological Society of San Diego by contacting Dr. Oliver Ryder at: oryder@ucsd.edu . None are available of the severely endangered Javan rhinoceros. An International Rhino Conference was held at the San Diego Zoo in 1991. It reviews many topics; a copy of the review of this conference can also be obtained from Dr. O. Ryder.
18) Other remarks – What additional Information is needed?
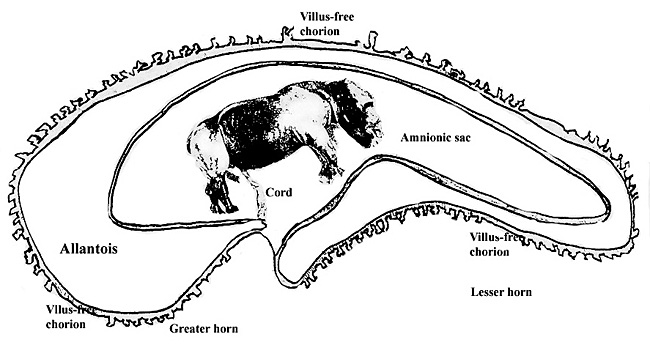
There are no studies on early implantation; information on the structure and length of umbilical cord is needed.
Acknowledgement
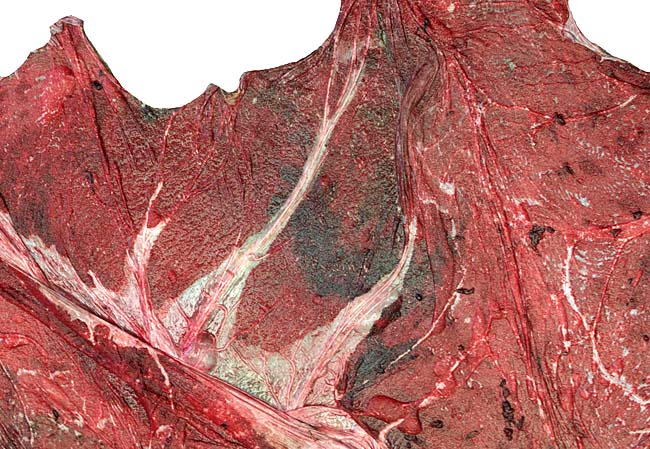
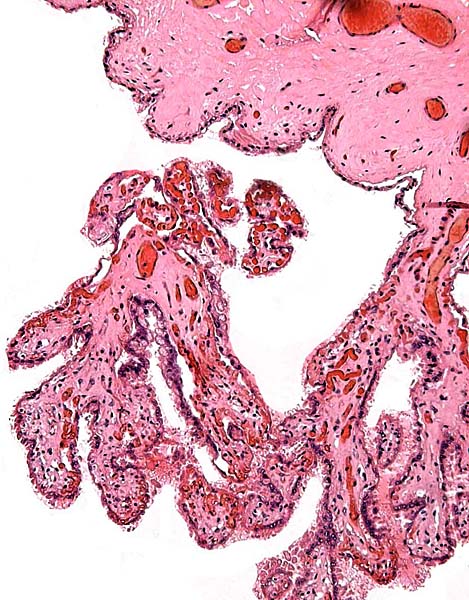
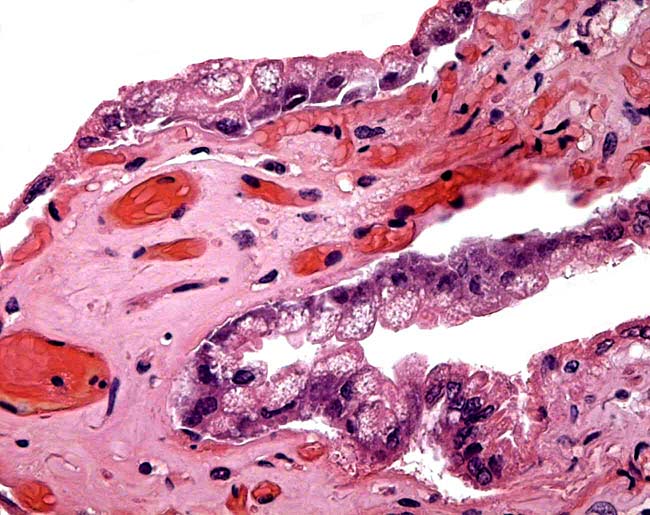
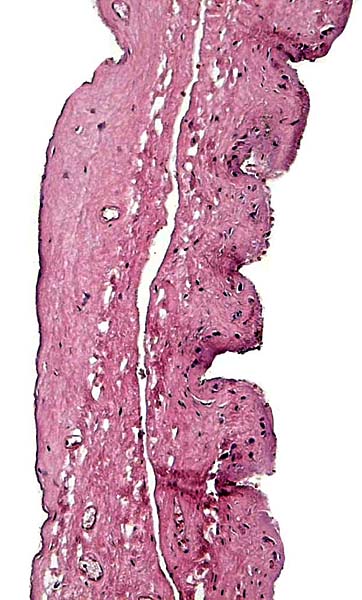
The animal photograph in this chapter comes from the Zoological Society of San Diego. The B&W photographs come from R. Montali at the National Zoo, Washington, DC.
References
Ali, S., Azfer, M.A., Bashamboo, A., Mathur, P.K., Malik, P.K., Mathur, V.B., Raha, A.K. and Ansari, S.: Characterization of a species-specific repetitive DNA from a highly endangered wild animal, Rhinoceros unicornis , and assessment of genetic polymorphism by microsatellite associated sequence amplification (MASA). Gene 228:33-42, 1999.
Anderson, E.C. and Rowe, L.W.: The prevalence of antibody to the viruses of bovine virus diarrhea, bovine herpes virus 1, rift valley fever, ephemeral fever and bluetongue and to Leptospira sp. in free-ranging wildlife in Zimbabwe. Epidemiol. Infect. 121:441-449, 1998.
Ashley, M.V., Melnick, D.J. and Western, D.: Conservation genetics of the black rhinoceros (Diceros bicornis), I. : Evidence from the mitochondrial DNA of three populations. Conserv. Biol. 4:71-77, 1990.
Benirschke, K. and Lowenstine, L.J.: The placenta of the rhinocerotidae. Verh. Ber. Erkr. Zootiere (Dresden). 37:15-23, 1995.
Brown, J.L., Bellem, A.C, Fouraker, M., Wildt, D.E and Roth, T.L.: Comparative analysis of gonadal and adrenal activity in the black and white rhinoceros in North America by noninvasive endocrine monitoring. Zoo Biol. 20:463-486 2001.
Chaplin, H. Jr., Malecek, A.C., Miller, R.E., Bell , C.E., Gray, L.S. and Hunter, V.L.: Acute intravascular hemolytic anemia in the black rhinoceros: hematologic and immunologic observations. Amer. J. Vet. Res. 47:13131-1320, 1986.
Clauss, M., Jessup, D.A., Norkus, E.B., Chen, T.C., Holick, M.F., Streich, W.J. and Dierenfeld, E.S.: Fat soluble vitamins in blood and tissues of free-ranging and captive rhinoceros. J. Wildl. Dis. 38:402-413, 2002.
Cunningham, J., Harley, E.H. and O'Ryan, C.: Isolation and characterization of microsatellite loci in black rhinoceros (Diceros bicornis). Electrophoresis 20:1778-1780, 1999.
Dierenfeld, E.S. du Toit, R. and Miller, R.E.: Vitamin E in captive and wild black rhinoceros (Diceros bicornis). J. Wildl. Dis. 24:547-550, 1988.
Fairbanks, V.F. and Miller, R.E.: Beta-globin chain hemoglobin polymorphism and hemoglobin stability in black rhinoceroses (Diceros bicornis). Amer. J. Vet. Res. 51:803-807, 1990.
Fischer-Tenhagen, C., Hamblin, C., Quandt, S. and Frolich, K.: Serosurvey for selected infectious disease agents in free-ranging black and white rhinoceros in Africa . J. Wildl. Dis. 36:316-323, 2000.
Galama, W.T., Graham, L.H. and Savage, A.: Comparison of fecal storage methods for steroid analysis in black rhinoceros (Diceros bicornis). Zoo Biol. 23:291-300, 2004.
Garnier, J.N., Holt, W.V. and Watson, P.F.: Non-invasive assessment of oestrous cycles and evaluation of reproductive seasonality in the female wild black rhinoceros (Diceros bicornis minor). Reproduction 123:877-889, 2002.
Garniera, J.N., Green, D.I., Pickard, A.R., Shaw, H.J. and Holt, W.V.: Non-invasive diagnosis of pregnancy in wild black rhinoceros (Diceros bicornis minor) by faecal steroid analysis. Reprod. Fertil. Dev. 10:451-458, 1998.
George, M. Jr., Chemnick, L.G., Cisova, D., Gabrisova, E., Stratil, A. and Ryder, O.A.: Genetic differentiation of white rhinoceros subspecies: diagnostic differences in mitochondrial DNA and serum proteins. In, Proc. Intern. Conference on Rhinoceros Biology and Conservation, San Diego , CA 1991, pp. 105-113.
Gotch, A.F.: Mammals – Their Latin Names Explained. Blandford Press, Poole , Dorset , 1979.
Grant, J.B., Brown, D.L. and Dierenfeld, E.S.: Essentially fatty acid profiles across diets and browse of black rhinoceros. J. Wildl. Dis. 38:132-142, 2002.
Griner, L.A. : Pathology of Zoo Animals. Zoological Society of San Diego , San Diego , California , 1983.
Groves , C.P.: Phylogeny of the living species of Rhinoceros. Z. zool. System. Evol. 21:293-313, 1983.
Hayssen, V., van Tienhoven, A. and van Tienhoven, A.: Asdell's Patterns of Mammalian Reproduction: a Compendium of Species-specific Data. Comstock/Cornell University Press, Ithaca , 1993.
Hindle, J.E., Mostl, E. and Hodges, J.K.: Measurement of urinary oestrogens and 20 alpha-dihydroprogesterone during ovarian cycles of black (Diceros bicornis) and white (Ceratotherium simum) rhinoceroses. J. Reprod. Fertil. 94:237-249, 1992.
Houck, M.L., Ryder, O.A., Kumamoto , A.T. and Benirschke, K.: Cytogenetics of the rhinocerotidae. Verhandlungsbericht der Erkrankungen der Zootiere. # 37:25-32, 1995). Published by Tierpark Berlin . (This is appended here at the end).
Hungerford, D.A., Chandra, H.S. and Snyder, R.L.: Somatic chromosomes of a black rhinoceros (Diceros bicornis Gray 1821). Amer. Naturalist 101:357-358, 1967.
Jama, M., Zhang, Y., Aman, R.A. and Ryder, O.A.: Sequence of the mitochondrial control region, tRNA Thr , tRNA Pro and tRNA Phe genes from the black rhinoceros, Diceros bicornis . Nucleic Acid Res. 21:4392, 1993.
Jones, D.M.: The husbandry and veterinary care of captive rhinoceroses. Intern. Zoo Ybk. 19:239, 251, 1979.
Kapur, V., Prasanth, S.G., O'Ryan, C., Azfer, M.A. and Ali, S.: Development of a DNA marker by minisatellite associated sequence amplification (MASA) from the endangered Indian rhino (Rhinoceros unicornis). Mol. Cell Probes 17:1-4, 2003.
Keirans, J.E.: Dermatocentor rhinocerinus (Denny 1843) (Acari: Ixodida:ixodidae): redescription of the male, female and nymph and first description of the larva. Onderstepoort J. Vet. Res. 60:59-68, 1993.
Kenny, D.E.: Salmonella sp. Survey of captive rhinoceroses in U.S. zoological institutions and private ranches. J. Zoo Wildl. Med. 30:383-388, 1999.
Kenny, D.E., Baier, J. and Getzy, D.M.: Salmonellosis in captive black rhinoceros (Diceros bicornis). J. Zoo Wildl. Med. 28:307-311, 1997.
Knapp, S.E., Krecek, R.C., Horak, I.G. and Penzhorn, B.L.: Helminths and arthropods of black and white rhinoceroses in southern Africa . J. Wildl. Dis. 33:492-502, 1997.
Kock, N. and Kock, M.D.: Skin lesions in free-ranging black rhinoceroses (Diceros bicornis) in Zimbabwe . J. Zoo Wildl. Med. 21:447-452, 1990.
Kock, N., Morton, D. and Kock, M.: Reproductive parameters in free-ranging female black rhinoceroses (Diceros bicornis) in Zimbabwe . Onderstepoort J. Vet. Res. 58:55-57, 1991.
Kock, R.A., Mihok, S.R., Wambua, J., Mwanzia, J. and Saigawa, K.: Effects of translocation on hematologic parameters of free-ranging black rhinoceros (Diceros bicornis michaeli) in Kenya . J. Zoo Wildl. Med. 30:389-396, 1999.
Lance, V.A., Patton, M.L. and Hagey, L.R.: Identification of a series of C(21)O(2) pregnanes from fecal extracts of a pregnant black rhinoceros (Diceros bicornis). Steroids 66:875-881, 2001.
Maluf , N.S. : Renal morphology of the hook-lipped African rhinoceros, Diceros bicornis , Linnaeus. Amer. J. Anat. 190:245-265, 1991.
Maluf. N.S. Further studies on the kidney of the hook-lipped African rhinoceros, Diceros bicornis . Anat. Rec. 238:38-48, 1994.
Meiswinkel, R.: Afrotropical culicoides: a redescription of C. (Avaritia) kanagai Khamala & Kette, 1971, reared from elephant dung in the Kruger National Park, South Africa). Onderstepoort J. Vet. Res. 54:585-590, 1987.
Merenlender, A.M., Woodruff, D.S., Ryder, O.A., Kock, R. and Váhala, J.: Allozyme variation and differentiation in African and Indian rhinoceroses. J. Hered. 80:377-382, 1989.
Mihok, S., Olubayo, R.O. and Moloo, S.K.: Trypanosomiasis in the black rhinoceros (Diceros bicornis Linnaeus, 1758). Rev. Sci. Tech. 11:1169-1173, 1992.
Miller, R.E.: Veterinary Bibliography for Rhinoceros. A.A. Balkema Publ., Amsterdam, 1983.
Miller, R.E. and Boever, W.J.: Fatal hemolytic anemia in the black rhinoceros: Case report and a survey. J.A.V.M.A. 181:1228-1231, 1982.
Miller, R.E., Cambre, R.C., de Lahunta, A., Brannian, R.E., Spraker, T.R., Johnson, C. and Boever, W.J.: Encephalomalacia in three black rhinoceroses (Diceros bicornis). J. Zoo Wildl. Med. 21:192-199, 1990.
Morales, J.C. and Melnick, D.J.: Molecular systematics of the living rhinoceros. Mol. Phylogenet. Evol. 3:128-134, 1994.
Munson, L., Koehler, J.W., Wilkinson, J.E. and Miller, R.E.: Vesicular and ulcerative dermopathy resembling superficial necrolytic dermatitis in captive black rhinoceroses ( Diceros bicornis ). Vet. Pathol. 35:31-42, 1998.
Neiffer, D.L., Klein, E.C. and Wallace-Switalski, C.: Leptospira infection in two black rhinoceroses (Diceros bicornis). J. Zoo Wildl. Med. 32:476-486, 2001.
Nijhof, A.M., Penzhorn, B.L., Lynen, G., Mollel, J.O., Morkel, P., Bekker, C.P. and Jongejan, F.: Babesia bicornis sp. Nov. and Theileria bicornis sp. Nov.: tick-borne parasites associated with mortality in the black rhinoceros (Diceros bicornis). J. Clin. Microbiol. 41:2249-2254, 2003.
O'Brien, J.K. and Roth, T.L.: Post-coital sperm recovery and cryopreservation in the Sumatran rhinoceros (Dicerorhinus sumatrensis) and application to gamete rescue in the African black rhinoceros (Diceros bicornis). J. Reprod. Fertil. 118:263-271, 2000.
Olivier, A., Lane, E., Volkmann, D.H., Hofmeyr, M. and Stegmann, G.F.: Rectal prolapse associated with a healed pelvic fracture in a pregnant free-ranging African black rhinoceros (Diceros bicornis). Part 2: surgery and necropsy. J. S. Afr. Vet Assoc. 72:242-244, 2001.
Ott, J. E., McDonald, S.E., Robinson, P.T. and Wright, F.H.: Ulcerative stomatitis in a black rhinoceros (Diceros bicornis). AAZV Ann. Proceed. 68-71, 1982.
Paglia, D.E.: Acute episodic hemolysis in the African black rhinoceros as an analogue of human glucose-6-phosphate dehydrogenase deficiency. Amer. J. Hematol. 42:36-45, 1993.
Paglia, D.E. and Radcliffe, R.W.: Anthracycline cardiotoxicity in a black rhinoceros (Diceros bicornis): evidence for impaired antioxidant capacity compounded by iron overload. Vet. Pathol. 37:86-88, 2000.
Paglia, D.E., Valentine, W.N., Miller, R.E., Nakatani, M. and Brockway, R.A.: Acute intravascular hemolysis in the black rhinoceros: Erythrocyte enzymes and metabolic intermediates. Amer. J. Vet. Res. 47:1321-1325, 1986.
Paglia, D.E., Kenny, D.E., Dierenfeld, E.S. and Tsu, I.H.: Role of excessive maternal iron in the pathogenesis of congenital leukoencephalomalacia in captive black rhinoceroses (Diceros bicornis). Amer. J. Vet. Res. 62:343-349, 2001.
Pessier, A.P., Munson, L. and Miller, R.E.: Oral, nasal, and cutaneous eosinophilic granulomas in the black rhinoceros (Diceros bicornis): a lesion distinct from superficial necrolytic dermatitis. J. Zoo Wildl. Med. 35:1-7, 2004.
Radcliffe, R.W., Paglia, D.E. and Couto, C.G.: Acute lymphoblastic leukemia in a juvenile southern black rhinoceros (Diceros bicornis). J. Zoo Wildl. Med. 31:71-76, 2000.
Radcliffe, R.W., Eyres, A.I., Patton, M.L., Czekala , N.M. and Emslie, R.H.: Ultrasonographic characterization of ovarian events and fetal gestational parameters in two southern black rhinoceros (Diceros bicornis) and correlation to fecal progesterone. Theriogenology 55:1033-1049, 2001.
Ryder, O.A., ed.: Rhinoceros Biology and Conservation. Zoological Society of San Diego , 1993 (Proc. of Conference 1991).
Schaffer, N., Bryant, W., Agnew, D., Meehan, T. and Beehler, B.: Ultrasonographic monitoring of artificially stimulated ejaculation in three rhinoceros species (Ceratotherium simum, Diceros bicornis, Rhinoceros unicornis). J. Zoo Wild. Med. 29:386-393, 1998.
Schaffer, N.E., Foley, G.L., Gill, S. and Pope, C.E.: Clinical implications of rhinoceros reproductive tract anatomy and histology. J. Zoo Wildl. Med. 32:31-46, 2001.
Schaurte, W.T.: Beiträge zur Kenntnis des Gebisses und Zahnbaues der afrikanischen Nashörner. Säugetierkundl. Mitt. 14:327-341, 1966.
Schwarzenberger, F., Francke, R. and Goltenboth, R.: Concentration of faecal immunoreactive progestagen metabolites during the oestrous cycle and pregnancy in the black rhinoceros (Diceros bicornis). J. Reprod. Fertil. 98:285-291, 1993.
Silberman, M.S. and Fulton , R.B.: Medical problems of captive and wild rhinoceros – a review of the literature and personal experiences. J. Zoo Anim. Med. 10:6-16, 1979.
Stegmann, G.F., Hofmeyr, M., Olivier, A., Lane, E. and Volkmann, D.H.: Rectal prolapse associated with a healed pelvic fracture in a pregnant free-ranging African black rhinoceros (Diceros bicornis). Part 1: anaesthesia. J. S. Afr. Vet. Assoc. 72:239-241, 2001.
Swart, M.K., Ferguson , J.W., du Toit, R. and Flamand, J.R.: Substantial genetic variation in southern African black rhinoceros (Diceros bicornis). J. Hered. 85:261-266, 1994.
Swart, M.K.J. and Ferguson , J.W.H.: Conservation implications of genetic differentiation in Southern African populations of black rhinoceros (Diceros bicornis). Conserv. Biol. 11:79-83, 1997.
Tougard, C., Delefosse, T., Hanni, C. and Montgelard, C.: Phylogenetic relationships of the five extant Rhinoceros species (Rhinocerotidae, Perissodactyla) based on mitochondrial cytochrome b and 12S rRNA genes. Mol. Phylogenet. Evol. 19:34-44, 2001.
Trifonov, V., Yang, F., Ferguson-Smith, M.A. and Robinson, T.J.: Cross-species chromosome painting in the Perissodactyla: delimitation of homologous regions in Burchell's zebra (Equus burchellii) and the white (Ceratotherium simum) and black rhinoceros (Diceros bicornis). Cytogenet. Genome Res. 103:104-110, 2003.
Turner, J.W. Jr., Tolson, P. and Hamad, N.: Remote assessment of stress in white rhinoceros (Ceratotherium simum) and black rhinoceros (Diceros bicornis). J. Zoo Wildl. Med. 33:214-221, 2002.
Weber, B.W., Paglia, D.E. and Harley, E.H.: Elevated free tyrosine in rhinoceros erythrocytes. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 138:105-109, 2004.
Woods, R., Blyde, D.J., Seaman, J.T. and Thorne, A.H.: Fungal pneumonia in a captive black rhinoceros. Austral. Vet. J. 77:717-719, 1999.
|
One female D. bicomis individual, (#360), was found to have a diploid number of 2n 85 because of an additional large submetacentric chromosome. Analysis of the 0-bands revealed a trisomy X chromosomal complement. Presumably the additional X did not have any deleterious effects because of X inactivation (LYON , 1962). This animal died at age 2.5 years. Autopsy findings gave no indications of anomalies or untoward effects of trisomy X. Diffuse encephalopathy was deemed to be the result of an unknown toxin or nutritional deficiency (Eric Miller, personal communication). The dam, (#267), of this individual was sampled and revealed a normal 2n 84 karyotype. The sire was not sampled directly, but karyotype analyses of his paternal grandparents, (#124 and #125), revealed normal chromosomal complements.
Two other female D. bicomis individuals, that were wild-caught and presumably unrelated, had normal diploid numbers, but showed very large heterochromatic areas in the p-arm of one chromosome. In one animal, (#293), the additional heterochromatin resulted in a submetacentric element similar in size to the X chromosomes. Due to the quality of the G-banding it was not possible to determine which chromosome was affected. In the other animal, (#188), a metacentric chromosome was created by the addition of a large heterochromatic arm. Q-banding showed a metacentric chromosome in which the q-arm paired with that of a subtelocentric autosome.
This study illustrates the importance of chromosomal analyses in support of animal health and conservation management programs. Further comparative studies need to be conducted on chromosomal polymorphisms which may distinguish different geographic populations of D. bicomis . In addition, the numerical polymorphism noted in the critically endangered northern white rhinoceros ( C. s. cottoni ) should be monitored through additional study of captive specimens and, where feasible, of wild populations.
Acknowledgements
The authors wish to thank Ken Kelley, Grace Magee, Suellen Charter and Reneê Cabrera for their assistance and cooperation. Appreciation is extended to Steve Kingswood, Dr. Valentine L. Vance and Dr. E. Ann 0akenfull for their critical review of the manuscript and to Dr. Mike H. Jurke for translation of the summary. Clonetics Corporation generously provided the necessary growth medium for the fibroblast cultures.
Summary
Cytogenetics of the Rhinocerotidae
Cytogenetic studies were conducted on 113 rhinoceroses, representing four of the five extant species. Karyotype analyses revealed a modal diploid number of 82 chromosomes for Ceratotherium simum, Rhinoceros unicomis, Dicerorhinus sumatrensis and 84 chromosomes for Diceros bicomis . Comparison of the autosomes of the four species revealed differences in centromere location, ranging from all acrocentric elements in D. sumatrensis to mostly submetacentric elements in D. bicomis. The X chromosome was identified as a large submetacentric element in all four species. The X chromosome of D. sumatrensis was distinct from the other three species because of an addition of heterochromatic material in the long arm.
Abnormal chromosomal complements were identified in three C. simum and three D. bicomis including one trisomy X individual. The diploid number of D. sumatrensis is presented for the first time, as well as the first banding studies of D. sumatrensis, R. unicornis and D. bicornis.
Zusammenfassung
CYTOGENETIK DER RHINOCEROTIDAE
Wir berichten über cytogenetische Untersuchungen an 113 Nashömem von vier der fünf bestehenden Nashomarten. Die Chromosomenzahl (“diploide Zahl" “2n”) beträgt 82 Chromosomen in den folgenden drei Arten: Ceratotherium simum, Rhinoceros unicornis und Dicerorhinus sumatrensis. Hingegen hat Diceros bicomis 84 Chromosomen. Wenn man die Autosomen dieser Arten vergleicht, so findet man eine unterschiedliche Lokalisation der Zentromeren, von ausschliesslich akrozentrischen Autosomen in D. sumatrensis zu hauptsächlich submetazentrischen E!ementen im D. bicomis. Das X-Chromosom ist ein grosses submetazentrisches Element in allen Arten. Das X-Chromosom von D. sumatrensis unterscheidet sich allerdings von den anderen Arten durch ein zusätzliches Stück von Heterochromatin im langen Arm. Chromosomenabnormalitäten wurden in je drei Nashömem von D. simum und von D. bicornis gefunden, einschliesslich einer Trisomie des X-Chromosoms. Dieser Bericht stellt die erste Beschreibung der diploiden Chromosomenzahl des D. sumatrensis dar; ebenso präsentieren wir die ersten gebandeten Chromosomen von D. sumatrensis, R. unicomis und von D. bicomis.
Résumé
La cytogénetique des rhinocérotidae
Nous avons effectué des examens cytogénétiques sur 113 rhinoceros ayant represente quatre des cinq espèces existantes. Le nombre de chromosomes (“chiffre diploide" "2n”) se chiffre a 82 chromosomes pour les trois espèces qui suivent: Ceratotherium simum, Rhinoceros unicornis et Dicerorhinus sumatrensis . Diceros bicomis par contre, a 84 XX chromosomes. La comparaison des autosomes de ces espèces révèle une localisation differente des centromères , a partir d'autosomes exclusivement acrocentriques dans l'espèce D. sumatrensis jusqu'aux elements essentiellement submétacentriques dans D. bicomis . Le chromosome X est un grand élément submetacentrique dans toutes les espèces. Cependant, le chromosome X de D. sumatrensis se distingue des autres espèces par un élement additionnel d'héterochromatine au bras long. Nous avons observe des anomalies de chromosomes dans trois specimens de C. simum et dans trois de D. bicornis ainsi qu'une trisomie du chromosome X. Ce rapport présente pour Ia premiere fois une description du chiffre diploide des chromosomes de D. sumatrensis et fait etat des bandes chromosomales de D. sumatrensis, de R. unicomis et de D. bicomis.
References
AMATO, G.D:, ASHLEY, M. and J. GATESY (1993): In, Rhinoceros biology and conservation (O. A. Ryder, ed). Zoological Society of San Diego , San Diego , 114-122.
HANSEN, KM. (1976): Q-bands of some chromosomes of the white rhinoceros. Hereditas 82, 205-208.
HEINICHEN, I. G. (1967): Karyotype of Ceratotherium simum s/mum and Equus zebra zebra. A preliminary note. J. S. Afr. Vet. Med. Assoc.38 247-248.
HEINICHEN, I.G. (1970): Karyologica/ studies on southern African Perissodactyla. Koedoe 13, 51-108.
HOUCK, M.L., RYDER, O.A., VAJALA, J., KOCK, R.A., and J.E. OOSTERHUIS (1993): Diploid chromosome number and chromosomal variation in the white rhinoceros (Ceratotherium simum). J. Hered. 85, 30-34.
HSU, T.C. and K. BENIRSCHKE (1973): An atlas of mammalian chromosomes. Vol. 7. Berlin : Springer- Ver/ag: folios 339-340.
HUNGERFORD, D.A., CHANDRA, H.S., and R.L. SNYDER (1967): Somatic chromosomes of a black rhinoceros (Diceros b/corn/s Gray 1821). Am. Nat. 101, 357-358.
GROOMBRIDGE, B., (Ed.) (1993) 1994 IUCN red list of threatened animals. !UCN Gland , Switzerland and Cambridge , UK .
GROVES , C.P. (1983): Phylogeny of the living species of rhinoceros. Z. Zoo!. Syst. Evolutionforsch. 21, 293-313.
LYON , M.F. (1962): Sex chromatin and gene action in the mammalian X-chromosome. Am. J. Hum. Genet. 14, 135-148.
MORALES, J.C. and D.J. MELNICK (1994): Molecular systematics of the living rhinoceros. Molec. Phylogen. and Evol. 128-134. OHNO, S. (1967): Sex chromosomes and sex-linked genes., Berlin/Heidelberg/ New York : Springer-Verlag.
PARIS CONFERENCE (1972): Standardization in human cytogenetics. Birth defects: Original art ser. VIII, No. 7. The National Foundation, New York .
PATHAK, Sand. D. STOCK (1974): The X chromosome of mammals: Karyological homology as revealed by banding techniques. Genetics 7_8, 703-714.
PENNY, M. (1988): Rhinos: Endangered Species. Facts on File Publications, New York .
PROTHERO, D.R., MANNING, E. and C.B. HANSSON (1986): The phylogeny of the Rhinocerotoidea (Mammalia, Perissodactyla.) Zoo! J. Linn. Soc. 87, 341-366.
RYBAK, J., THARAPEL, A., ROBINETT, S., GARCIA, M., MARK!NEN C., and M. FREEMAN (1982): A simple reproducible method for prometaphase chromosome analysis. Hum. Gen. 60, 328-333.
RYDER, O.A., HOUCK, M.L., and A.T. KUMAMOTO (1987): Rhinoceros genetics: the state-of-the-art and application to conservation measures. American Association of Zoological Parks Annual Proceedings, 710-714.
SEABRIGHT, M. (1971): A rapid technique for human chromosomes. Lancet 2, 971-9 72.
SUMNER, A. T. (1972): A simple technique for demonstrating centromeric heterochromatin. Exp. Cell Res. L5, 304-306.
WURSTER, D.H. and K. BENIRSCHKE (1968): The chromosomes of the great Indian rhinoceros. Experientia 24. 511.
YU, R.L., ARONSON, M.M. and WW. NICHOLS (1981): High-resolution bands in human fibroblast chromosomes induced by actinomycin D. Cytogenet. Cell Genet. 31, 111-114.
Address of authors: Marlys L. Houck, Center for Reproduction of Endangered Species, Zoological Society of San Diego , P. 0. Box 551 , San Diego , C.A. 92112-0551 (USA).
|