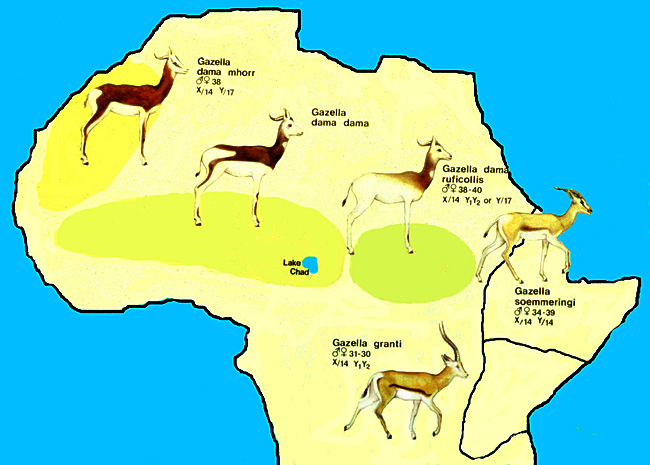
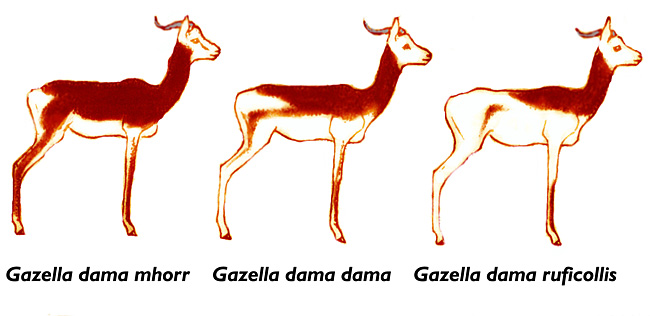
2) General Gestational Data
The length of gestation is 5 months according to Rode (quoted by Mentis, 1972). They also indicated that longevity is between 9 and 12 years, but Jones (1993) gave 17+ years as longevity. Gestation lasts 200 days according to Furley, (1986) and one young is generally born. Numerous animals have been born from the small initial stock at San Diego , and in other institutions as well.
3) Implantation
This pregnant animal from which the specimen comes died at San Diego 's Wild Animal Park for no known reasons. The animal had done well in the past. The interesting feature of this young gestation is that the corpus luteum was in the right ovary but the fetus lay in the left uterine horn. Both horns contained placental cotyledons. The male fetus of this gestation weighed only 95 g and was 14.5 cm in CR length.
4) General Characterization of the Placenta
This is a polycotyledonary chorio-epithelial placenta whose cotyledons were located in both uterine horns, even though the fetus was confined to the left uterine horn. The left horn had 58 cotyledons; the right horn had 34 (total 92) cotyledons. They were obviously very small, maximally 2 x 1 cm in diameters.
|
|
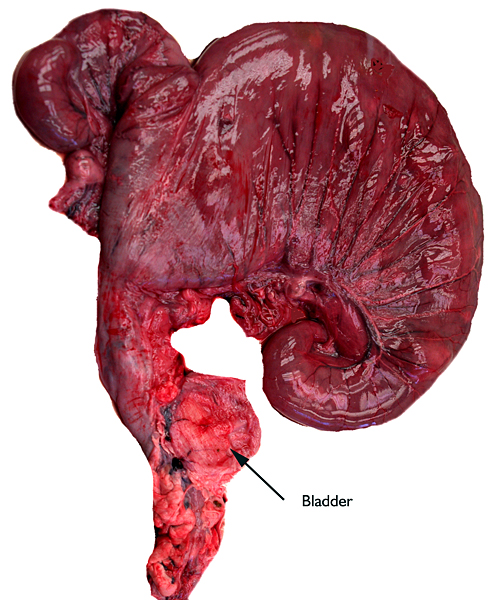 |
Gross specimen of pregnant uterus with fetus in the left horn.
|
| |
|
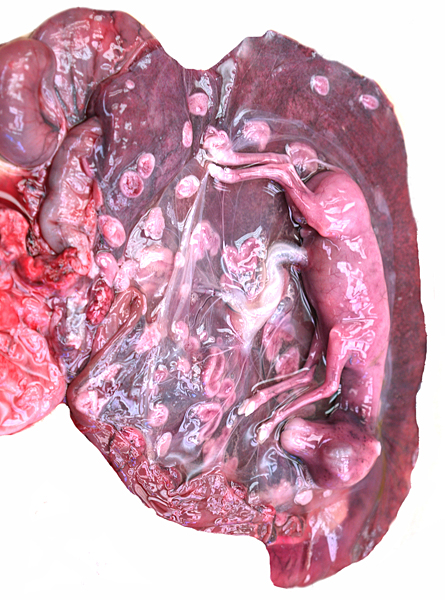 |
Opened uterus with fetus in left horn. Short umbilical cord.
|
| |
|
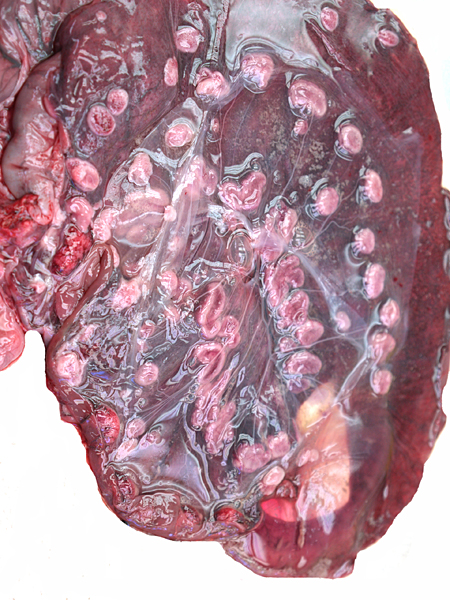 |
After the removal of the fetus, the concave placental; cotyledons are apparent.
|
5) Details of fetal/maternal barrier
The “barrier” is typical of that seen in most ungulates. The villous surfaces interdigitate with maternal septa. The trophoblast is single-layered and has numerous binucleate cells. There is no invasion of the maternal structures. In contrast to many other ungulate placentas, there is no pigmentation and no evidence of a “hematophagous” organ, at least at this stage of development.
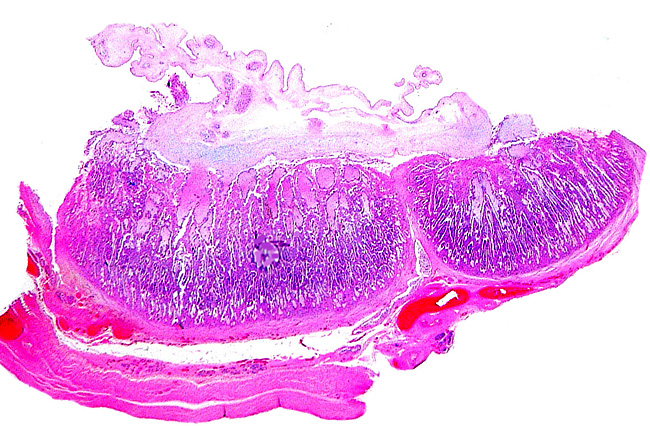 |
Two neighboring cotyledons.
|
| |
|
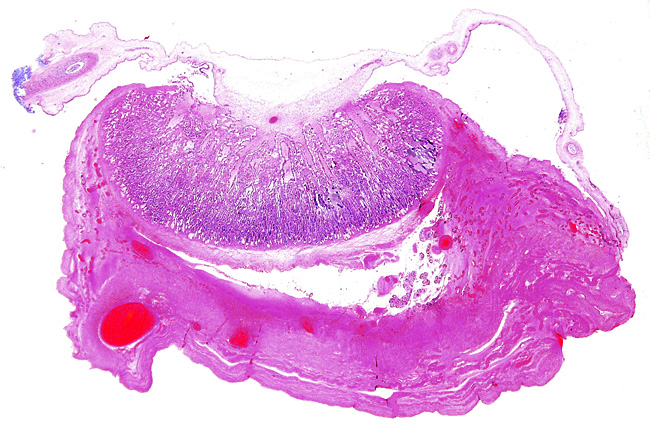 |
One of the smaller implanted cotyledons slightly separated from uterus.
|
| |
|
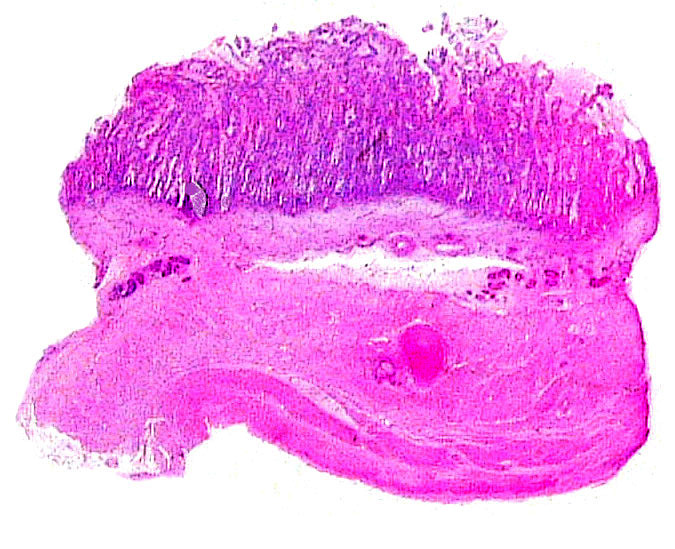 |
Cotyledon from “empty” horn.
|
| |
|
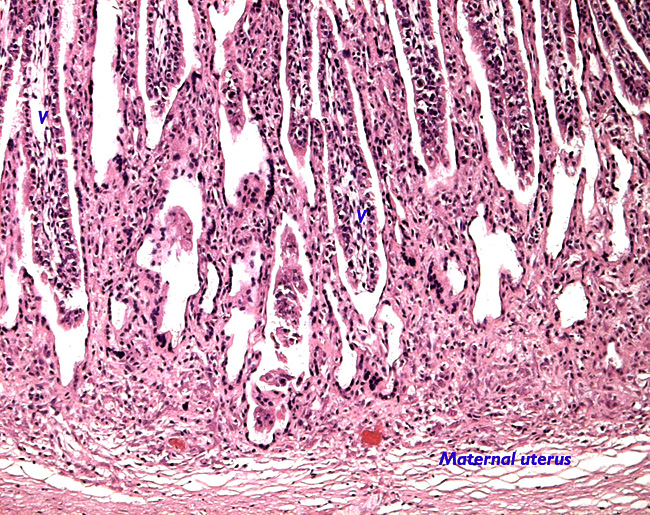 |
Uterine implantation of cotyledon.
|
| |
|
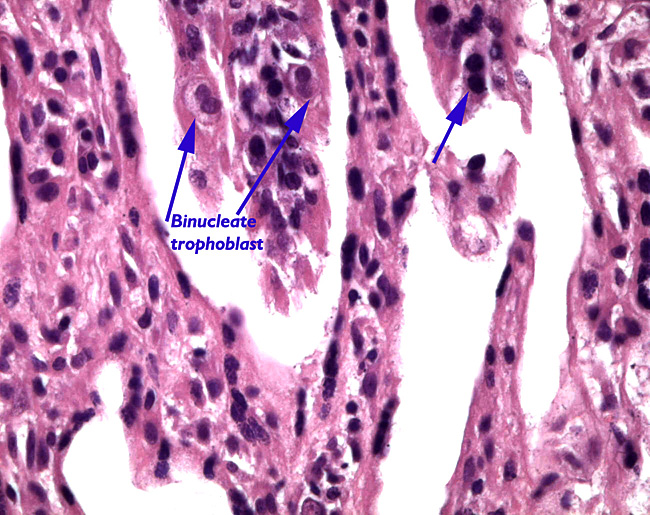 |
Binucleate trophoblast at arrows.
|
| |
|
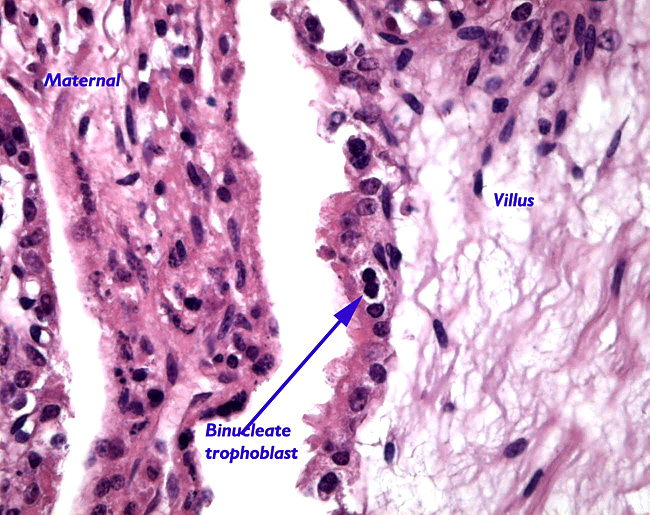 |
Maternal septum (left) and villus with binucleate cell (right).
|
6) Umbilical cord
The umbilical cord was essentially comprised only of the amnionic portion and measured 5 cm in length. It contained four vessels and the allantoic duct. Its surface was covered by numerous minute foci of squamous metaplasia. There were no twists. The allantoic portion was virtually not extant, as the allantoic vessels split immediately at the site of allantoic membrane attachment.
|
|
 |
Umbilical cord with four vessels and central (high) allantoic duct. Fine foci of squamous metaplasia are seen on cord surface.
|
| |
|
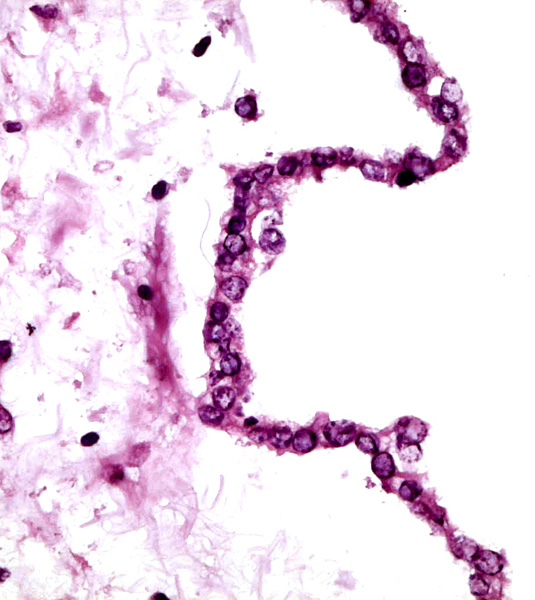 |
Epithelium of allantoic duct in the umbilical cord.
|
| |
|
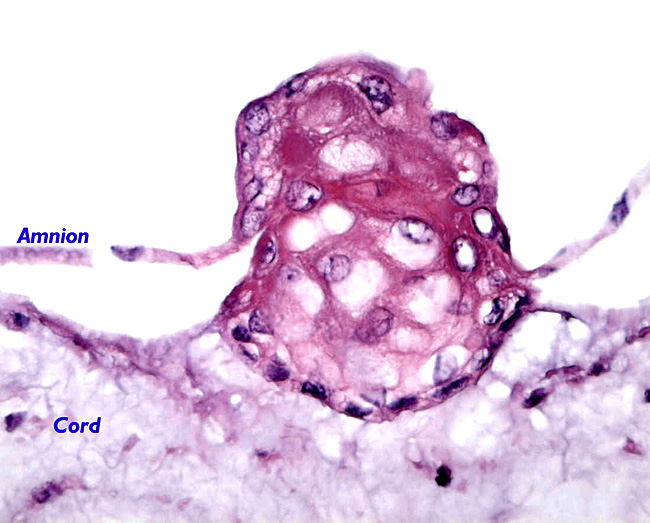 |
Focus of squamous metaplasia from the cord surface.
|
| |
|
7) Uteroplacental circulation
There are no studies.
8) Extraplacental membranes
The allantoic epithelium is single-layered and very thin; its connective tissue has numerous vessels. The yolk sac is atrophied. The amnion has a single-layered squamous epithelium and is avascular. There is no decidua capsularis.
9) Trophoblast external to barrier
There is no infiltration of trophoblast into the endometrium.
10) Endometrium
No decidua is formed.
11) Various features
No unusual features are present.
12) Endocrinology
The endocrine features of the reproductive cycles in Mhorr gazelles (!) were detailed by Pickard et al. (2001) from fecal specimens. The response by males to changing estrogen levels of females was studied by Pickard et al. (2003). Neumann et al. (2002) determined fecal progesterone levels in captive animals, concentrating primarily on the stability of the hormone after freezing etc.
13) Genetics
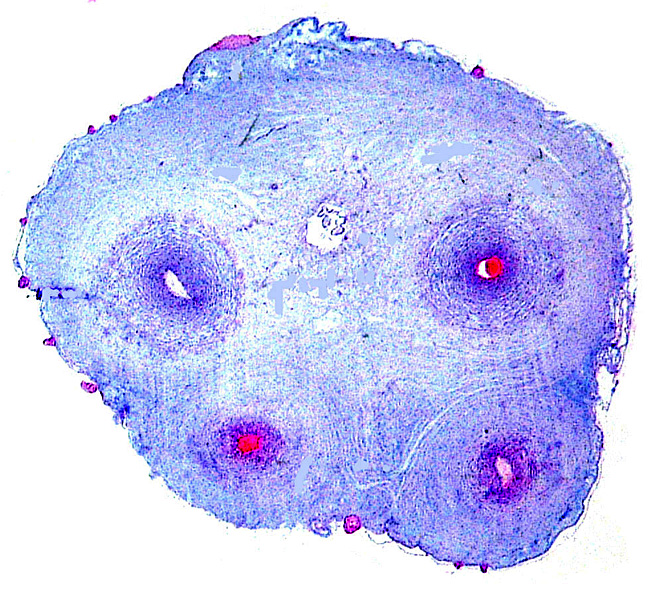
The first karyotype of a male Dama gazelle had 38 chromosomes, 22 metacentrics and 16 acrocentrics; the sex chromosomes were then unknown (Wurster & Benirschke, 1968). It must be admitted, however, that this specimen was most likely a red-necked gazelle, not the nominate form. Vassart et al. (1995) also depicted a male Dama gazelle karyotype with 2n=38 (see next figure). Subsequently, many additional animals were studied in San Diego and Effron et al. (1976) were able to state that G. dama had 2n=39 or 40 in males, and 2n=38 or 40 in females, all wit X/A translocation.
Altogether we had the following karyotypes:
9 females with 2n=38
1 female with 2n=39
1 female with 2n=40
1 male with 2n-38
4 males with 2n=39
1 male with 2n=40
In 1993, however, Vassart et al. (1993) published a Dama gazelle karyotype with 2n=34. It came from a female animal at Taif, but the precise ultimate origin of this animal was, again, unknown. Its phenotype also differed much from the D. ruficollis stock kept at San Diego . Its rump is deeply dark colored and is thus very similar to the picture of the Sömmerring's gazelle in its original description (see chapter on Sömmerring gazelle). It is thus apparent that there karyotypic or phenotypic problems with this species, not unlike G. sömmerringi.
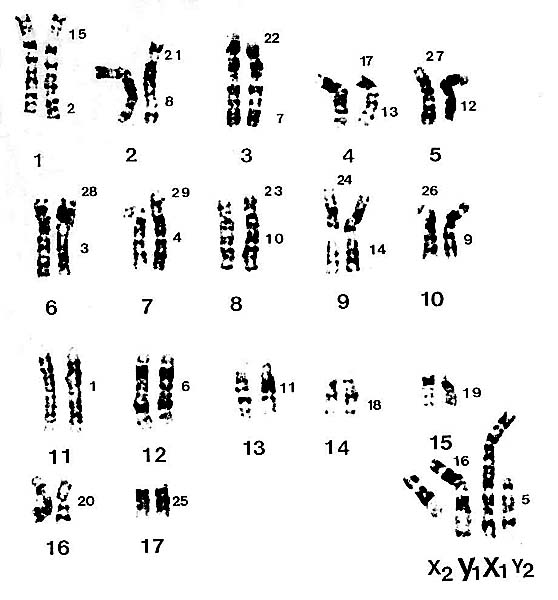 |
Karyotype of male specimen from San Diego , as presented by Vassart et al. (1995). Chromosomes are numbered according to cattle standard. The X/A translocation is apparent.
|
| |
|
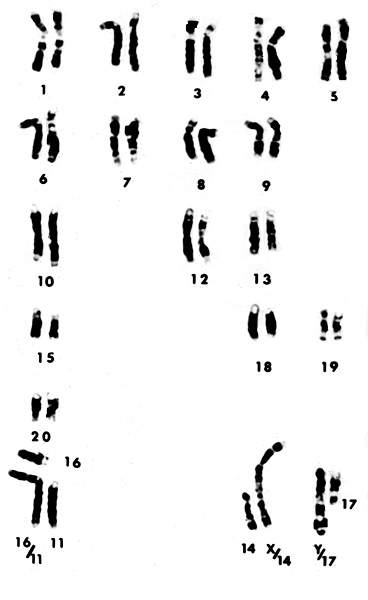 |
Karyotype of male Dama gazelle with 2n=39.
|
| |
|
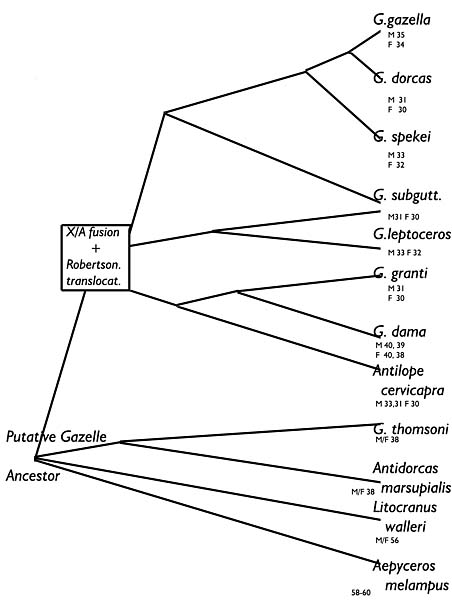 |
This is my suggestion for a tree of antelopes/gazelles of Africa. It is related to the many results of our chromosomal studies. The bottom four species do not possess the X/A translocation of the animals above. Branching points indicate additional Robertsonian fusions.
|
| |
|
14) Immunology
I am not aware of any immunological studies in this species.
15) Pathological features
Griner (1983) summarized his experience with fifteen deaths of the 48 animals born in San Diego . Trauma and neonatal deaths due to neglect were the principal causes of mortality. Bluetongue infection occurred in another. An adrenal adenoma was described by Schmidt & Fletcher (1979), and Martin et al. (1985) described a cementoblastoma in a 6 year old animal whose jaw had been swollen for two years. After removal, the animal did well.
16) Physiologic data
Hematologic values and some biochemical information on several Nanger species are available from a publication by Casado et al. (1991). Schumacher et al. (1997) evaluated the cardiac and pulmonary sequelae following administration of the immobilizing agent carfentanil. Gomendio et al. (2000) compared the ejaculated semen of three gazelle species and showed that the ejaculates were negatively influenced by the degree of inbreeding. Sperm collection, cryopreservation and thawed sperm evaluation were evaluated by Garde et al. (2003).
17) Other resources
Numerous samples of frozen fibroblasts are available from CRES at the Zoological Society of San Diego by contacting Dr. Oliver Ryder at oryder@ucsd.edu .
18) Other remarks – What additional Information is needed?
Mature placentas have not yet been available, nor are their weights.
Acknowledgement
The animal photographs in this chapter come from the Zoological Society of San Diego and my personal collection.
References
Casado, A., de la Torre, R., Lopez-Fernandez, E. and Ruiz del Castillo, B.: Hematologic and biochemical observations in Gazella dama, G. dorcas and G. cuvieri. Comp. Biochem. Physiol. B. 99:637-640, 1991.
Effron, M., Bogart, M.H., Kumamoto , A.T. and Benirschke, K.: Chromosome studied in the mammalian subfamily Antilopinae . Genetica 46:419-444, 1976.
Furley, C.W.: Reproductive parameters of African gazelles: Gestation, first fertile matings, first parturition and twinning. Afr, J. Ecol. 24:121-128, 1986
Garde, J.J., Soler, A.J. and Cassinello, J., Crespo, C., Malo, A.F., Espeso, G., Gomendio, M. and Roldan, E.R.: Sperm cryopreservation in three species of endangered gazelles (Gazella cuvieri, G. dama mhorr, and G. dorcas neglecta). Biol. Reprod. 69:602-611, 2003.
Gomendio, M., Cassinello, J. and Roldan, E.R.: A comparative study of ejaculate traits in three endangered ungulates with different levels of inbreeding: fluctuating asymmetry as an indicator of reproductive and genetic stress. Proc. Royal Soc. London B Biol. Sci. 267:875-882, 2000.
Griner, L.A. : Pathology of Zoo Animals. Zoological Society of San Diego , San Diego , California , 1983.
Jones, M.L.: Longevity of ungulates in captivity. Int. Zoo Yearb. 32:159-169, 1993.
Lange, J.: Ein Beitrag zur systematischen Stellung der Spiegelgazellen (Genus Gazella Blainville, 1816 Subgenus Nanger Lataste, 1885). Z. Säugetierk. 36:1-18, 1971.
Martin, H.D., Turner, T., Kollias, G.V., Lin, S.L., Heard, D.J. and Jacobson, E.: Cementoblastoma in a Dama gazelle. J. Amer. Vet. Med. Assoc. 187:1246-1247, 1985.
Mentis, M.T.: A review of some life history features of the large herbivores of Africa . The Lammergeyer 16:1-89, 1972.
Neumann, G., Gottschalk, J., Eulenberger, K. and Grun, E.: The stability of progesterone in feces of different wild animal species kept in a zoological garden. Dtsch. Tieraerztl. Wochenschr. 109:245-249, 2002 (in German).
Pickard, A.R., Abaigar, T., Green, D.I., Holt, W.V. and Cano, M.: Hormonal characterization of the reproductive cycle and pregnancy in the female Mohor gazelle (Gazella dama mhorr). Reproduction 122:571-580, 2001.
Pickard, A.R., Holt, W.V., Green, D.I., Cano, M. and Abaigar, T.: Endocrine correlates of sexual behavior in the Mhorr gazelle (Gazella dama mhorr). Horm. Behav. 44:303-310, 2003.
Rode, P.: Mammiferes Ongulés de l'Afrique Noire. Paris , 1943.
Schmidt, R.E. and Fletcher, K.C.: Adrenal cortical adenoma in a dama gazelle (Gazella dama). J. Wildl. Dis. 15:299-301, 1979.
Schumacher, J., Heard, D.J., Young, L. and Citino, S.B.: Cardiopulmonary effects of carfentanil in dama gazelles (Gazella dama). J. Zoo Wildl. Med. 28:166-170, 1997.
Vassart, M., Greth, A., Durand, V. and Cribiu, E.P.: An unusual Gazella dama karyotype. Ann. Genet. 36:117-120, 1993.
Vassart, M., Séguéla, A. and Hayes, H.: Chromosomal evolution in gazelles. J. Hered. 86:216-227, 1995.
Warhol, A. and Benirschke, K.: Vanishing Animals. Springer-Verlag , N.Y. 1986.
Wilson , D.E. and Reeder, D.A.M., eds.: Mammal Species of the World. A Taxonomic and Geographic Reference. 2 nd ed. Smithsonian Institution Press, Washington , DC , 1993.
Wurster, D.H. and Benirschke, K.: Chromosome studies in the superfamily Bovoidea. Chromosoma 25:152-171, 1968.
|