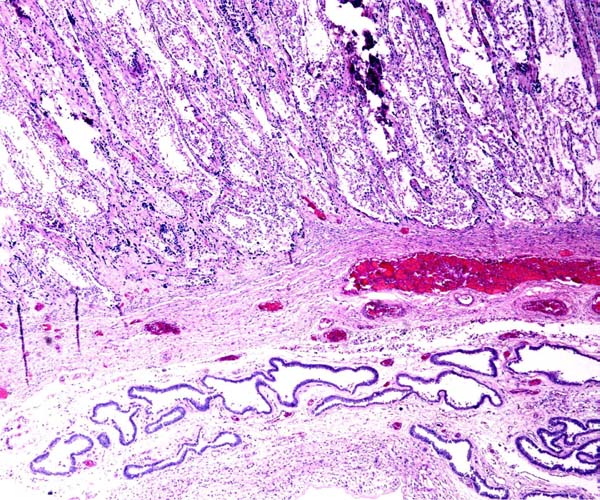
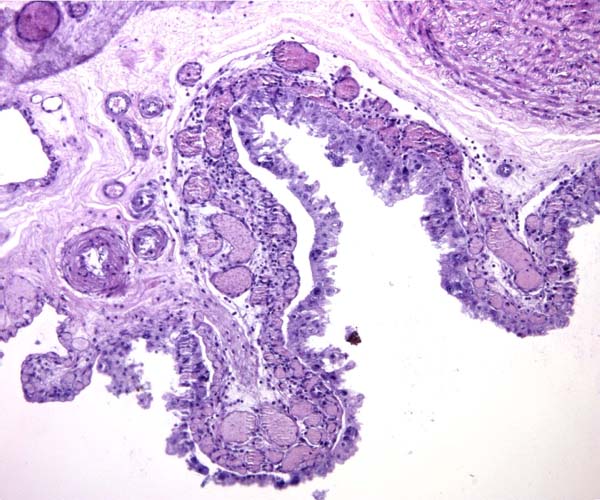
6) Umbilical cord
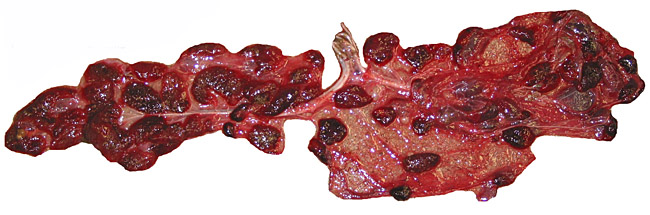
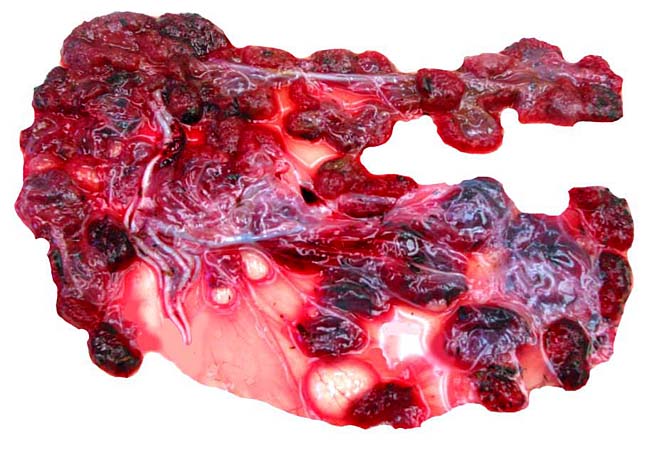
The umbilical cord contains 4 large blood vessels and a large allantoic duct. The umbilical cord is inserted in the mid-portion of the placenta; it has no spirals. Numerous smaller vessels are present in the cord, in addition to the principal blood vessels. Some of the smaller vessels surround the allantoic duct. The cord of the placenta here depicted was 6 cm long; the fetus was stillborn at term. The cord of a live-born obtained in 2004 was only 3 cm long. The allantoic duct is lined by urothelium.
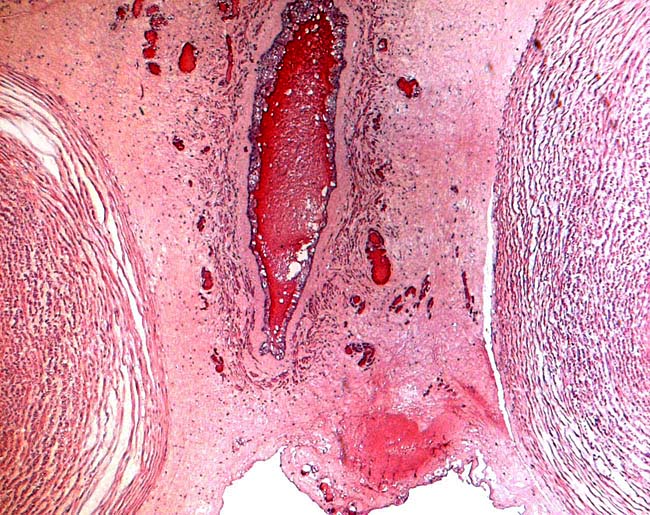 |
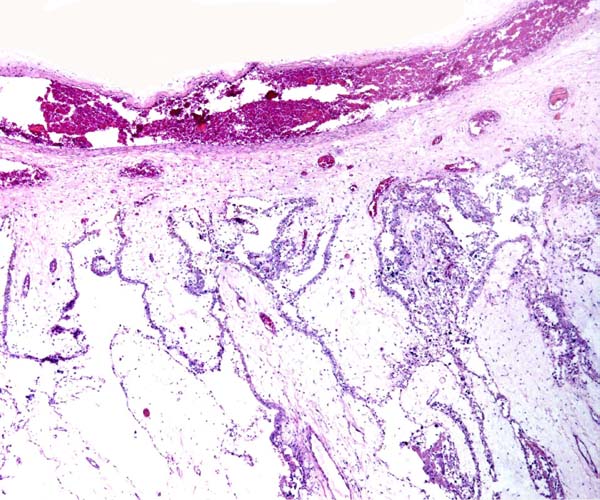
Central portion of umbilical cord near the placental surface. This shows the central allantoic duct with blood (Cesarean section-induced), allantoic vessels and musculature of duct.
|
7) Uteroplacental circulation
This has not been studied.
8) Extraplacental membranes
The allantoic sac has the characteristic vasculature; hippomanes have not been seen. The amnion has a single-layered flat epithelium. Vitelline remnants are not found at term.
9) Trophoblast external to barrier
There is none.
10) Endometrium
True decidual transformation of the endometrium does not occur.
11) Various features
Whether a subplacenta exists is unknown. No well-preserved implanted placentas have been available, nor have they been described in the literature.
12) Endocrinology
I know no endocrine studies.
13) Genetics
Dorcas gazelles have 31 chromosomes in the males, and 30 chromosomes in females. This species, as so many other Antilopinae, has a fusion between an autosome (the autosome is always the same element) and X-chromosome. Hence, the females have one less chromosome, but the DNA/genetic information is otherwise identical. An extensive chromosomal survey of these species was reported by Effron et al. (1976). From that analysis, the suggestion comes that there must have been one ancestor with this typical fusion of chromosomes that gave rise to most African gazellini. Hybrids between Gazella dorcas and Gazella gazella have been reported by Wahrman et al. (1973). Males were sterile, females were subfertile. Ralls et al. (1980) have discussed the inbreeding depression occurring in the highly productive herd in Washington.
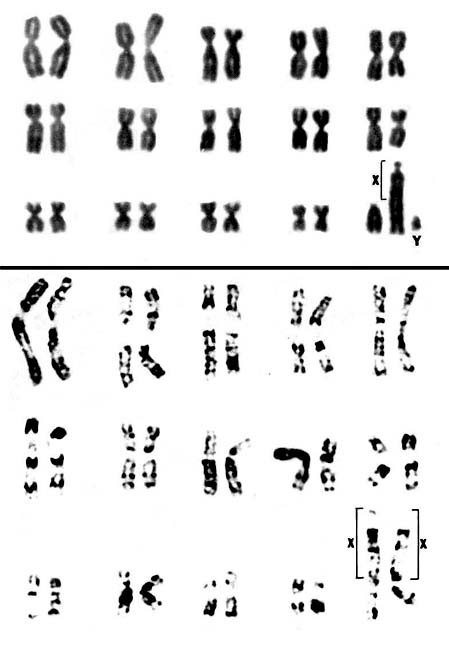 |
Chromosome karyotypes of male (above) and female Dorcas gazelles (from Hsu & Benirschke, 1974).
|
14) Immunology
There are no studies.
15) Pathological features
Griner (1983) found a myocardial infarct and aortic arteriosclerosis in an aged Dorcas gazelle. Kranz et al. (1984) reported that parasitic infections have been a consistent problem in captivity. Dittrich (1970) found the cause of deaths in captive Dorcas gazelles as follows: septicemia (0.1); pleurisy/hepatitis (1.1); gastroenteritis (1.0).
Haenichen et al. (2002) reported the unusual infection of a pregnant dorcas gazelle with an unusual form of algae, Chlorella, that causes the green granulomatous inflammation known as "Chlorellosis". The infection recurred and the animal was sacrificed. This infection has only rarely been reported in mammals (cattle, sheep, dromedary).
We have seen one gestation that ended with a set of conjoined twins. Therefore, it is possible that the occasional twins that are encountered in this species may be of monozygotic origin.
16) Physiologic data
No studies exist.
17) Other resources
The Zoological Society of San Diego in its CRES division has fibroblast cell lines in the "Frozen Zoo". They can be made available by Dr. Oliver Ryder at (oryder@ucsd.edu).
18) Other remarks - What additional Information is needed?
We need better information on the frequency of binucleate cells, the possible (likely) presence of "areolae" in the intercotyledonary region, and a description of the maternal/fetal interface of an implanted placenta. Endocrine studies are lacking.
Acknowledgement
I appreciate the consistent help of the pathologists at the San Diego Zoo in collecting this material.
References
Baharav, D.: Reproductive strategies in female mountain and Dorcas gazelles (Gazella gazella gazella and Gazella dorcas). J. Zool. 200:445-453, 1983.
Dittrich, L.: Beitrag zur Fortpflanzungsbiologie afrikanischer Antilopen im zoologischen Garten. Zool. Garten 39:16-40, 1970.
Effron, M., Bogart, M.H., Kumamoto, A.T. and Benirschke, K.: Chromosome studies in the mammalian subfamily Antilopinae. Genetica 46:419-444, 1976.
Griner, L.A.: Pathology of Zoo Animals. Zoological Society of San Diego. San Diego, California, 1983.
Haenichen, T., Facher, E., Wanner, G. and Hermanns, W.: Cutaneous chlorellosis in a gazelle (Gazella dorcas). Vet. Pathol. 39:386-389, 2002.
Hayssen, V., van Tienhoven, A. and van Tienhoven, A.: Asdell's Patterns of Mammalian Reproduction: a Compendium of Species-specific Data. Comstock/Cornell University Press, Ithaca, 1993.
Jope, M.: Meine Erfahrungen in der Aufzucht von Dorcasgazellen und Axis-und Schweinshirschen. D. Zool. Garten 49:374-376, 1908.
Kranz, K.R., Xanten, W.A. and Lumpkin, S.: Breeding history of the Dorcas gazelles Gazella dorcas at the National Zoological Park, 1961-1981. Int. Zoo Yearb. 23:195-203, 1984.
Nowak, R.M.: Walker's Mammals of the World. 6th ed. The Johns Hopkins Press, Baltimore, 1999.
Puschmann, W.: Zootierhaltung. Vol. 2, Säugetiere. VEB Deutscher Landwirtschaftsverlag Berlin, 1989.
Ralls, K., Brugger, K. and Glick, A.: Deleterious effects of inbreeding in a herd of captive Dorcas gazelle Gazella dorcas. Int. Zoo Yearb. 20:137-146, 1980
Slaugther, L.: Gestation period of the Dorcas gazelle. J. Mammal. 52:480-481, 1971.
Wahrman, J., Richler, C., Goitein, R., Horowitz, A. and Mendelssohn, H.: Multiple sex chromosome evolution, hybridization and differential X chromosome inactivation in gazelles. In: Jerusalem Chromosome Conference, and Chromosomes Today 42:434-435, 1973.
|